WARN-ID has been awarded supplements to fund two new exciting projects. The first project, led by the Broad Institute, will deploy their microfluidic platform, mCARMEN, to perform cost-effective surveillance of SARS-CoV-2 and other respiratory viruses at Kenema Government Hospital (KGH) in Sierra Leone. The second project, led by Scripps Research, will investigate rebound cases of SARS-CoV-2 after treatment with the oral antiviral Paxlovid.
mCARMEN deployment in Sierra Leone
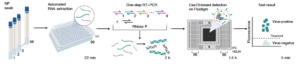
Schematic of the streamlined mCARMEN workflow
As the improved and updated version of CARMEN, mCARMEN will be utilized to dramatically expand surveillance capabilities for SARS-CoV-2 and other priority respiratory pathogens and to also serve as a catalog of endemic viral, bacterial, and parasitic pathogens. This project aims to deploy mCARMEN equipment and reagents at KGH, generate data on over 1,500 specimens from patients with respiratory and febrile illnesses, and prepare plans on how to further scale-up the project and increase data integration beyond the scope of the award. The teams at the Broad Institute and KGH aim to address the gap in undiagnosed febrile illness due to limited testing capacity, and create an effective solution for rapid testing and reporting by combining mCARMEN with WARN-ID data visualization platforms. Deploying mCARMEN as part of a holistic surveillance system in Sierra Leone will help lay the groundwork for a robust, sustainable public health infrastructure and serve as a model for viral surveillance systems in other resource-limited countries.
Paxlovid Study
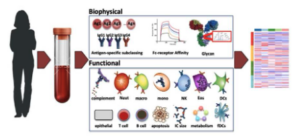
Systems Serology workflow
The project, led by Scripps Research aims to address the research gap in understanding the extent and characteristics of rebound SARS-CoV-2 after antiviral treatment. Paxlovid is the most prescribed oral antiviral for COVID-19, but there are no incidence or prevalence numbers on rebound cases following its five-day treatment course, despite multiple reports of such cases. In particular, the aim of this project is to investigate the incidence, prevalence, symptomatology, virus evolution, and immune correlates of COVID-19 cases that rebound after completion of Paxlovid treatment. Given the widespread use of oral antivirals for five days after acute COVID-19, understanding the incidence and symptomatology of Paxlovid rebound is essential. Understanding the epidemiology and pathophysiology of Paxlovid rebound would inform both policy and management decisions surrounding the prescribed treatment course, and would also illuminate the factors that can predict rebound SARS-CoV-2 after Paxlovid treatment.


